Local anti-infective agents and Preservatives
Contents
• Anti-infective
agents – classification
• Local
Anti-infective agents – Introduction & Classification
• Mode
of action of various Local Anti-infective agents
• Synthesis and specific uses of Furazolidone
• Preservatives
– Ideal characteristics & types
Learning Objectives
At the
end of this lecture, student will be able to
Describe the term “anti-infective agent”
Classify anti-infective agents
Enlist the ideal characteristics of Local anti-infective
agents (Germicides) & Preservatives
Classify Germicides & Preservatives
Describe the mode of action of Germicides
Introduction and Classification of anti-infectives
• Anti-infective
agents are those agents which are used in the treatment of infectious diseases
• Selective
toxicity is the main aim of modern “anti-infective therapy”
Classification:-
• Anti-infective
agents may be classified according to a variety of schemes, as the chemical
type of the compound the biological property and therapeutic indication
• A
combination of these classification schemes is used to classify the
anti-infective agents as
Classification of anti-infectives
Ø Local Anti-infective agents:-
• Alcohols – ethanol, isopropyl alcohol
• Phenols – p-chlorophenol, hexachlorophene,
resorcinol, hexyl resorcinol
• Halogen
– containing compounds- Iodine tincture, Halozone.
• Oxidizing
agents – Hydrogen peroxide, Benzoyl peroxide.
• Cationic
surfactants – Benzalkonium chloride, Cetyl pyridium chloride
• Dyes
– gentian violet, Methyene blue.
• Nitrogen
compounds – Nitrofurazone, Furazolidone.
• Mercury
compounds – Nitromersol and Thimerosal
Ø Preservatives :-
• p-hydroxy
benzoic acid derivatives – Methyl paraben, etc.,
• Miscellaneous
compounds as chlorobutanol, etc.,
Ø Anti-fungal agents :-
• Anti-fungal
antibiotics – Nystatin, Candicidin, Hamycin, Griseofulvin, Amphotericin – B.
• Synthetic
anti-fungal agents
• Substituted
imidazoles: Clotrimazole, Metronidazole, Ketoconazole.
• Miscellaneous
compounds- Zinc propionate, Sodium Caprylate, Tolnaftate.
Ø Urinary tract anti-infectives :-
• Quinolones
– Nalidixic acid, Norfloxacin, Ciprofloxacin, Perfloxacin
• Miscellaneous
– Nitrofurantoin.
Ø Anti-tubercular agents:-
• Synthetic
anti-tubercular agents- PAS, INH, Ethanbutol, Pyrazinamide, etc.,
• Anti-tubercular
antibiotics – Cycloserine, Rifampicin.
Ø Anti-viral agents:- as
• Amantadine
HCl, Acyclovir, Zidovudine
• Anti-AIDS
– Azathymidine, Suramin.
Ø Anti – protozoal agents:-
• Metronidazole,
Iodoquinol, Dimercaprol
Ø Anthelmintics:-
• Diethyl
Carbamazine, Mebendazole, Niclosamide
• Anti-scabious
& anti-pedicular agents:-
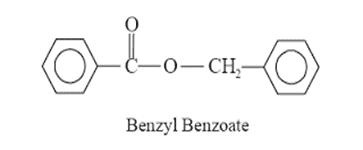
• Benzyl
benzoate, Diethyl toluamide, Lindane.
Benzyl Benzoate
Diethyl toluamide
Lindane
Local Anti-infective agents (Germicides)
There are two primary sub-types
Antiseptics:-
kill (-cidal) or prevent the growth of (-static) micro-organisms when applied
to living tissues.
Ø A
useful antiseptic must –
• Have
low toxicity so that it can be used directly on skin or wounds.
• Have
a rapid and sustained lethal action against micro-organisms.
• Have
low surface tension, so that it will spread into the wound.
• Have
the ability to retain activity in the presence of body fluids including pus.
• Be
non-irritating to tissues & non-allergic
• Lack
systemic toxicity when applied to the skin or mucous membrane.
• Have
no interference with the healing process of the wound.
Disinfectants:-
Ø A
disinfectant is an agent that prevents transmission of infection by the
destruction of pathogenic micro-organisms when applied to inanimate objects.
Ø An
ideal disinfectant:-
• Exerts
a rapid lethal action against all potentially pathogenic microorganisms &
spores.
• Have
good penetrating properties into organic matter.
• Should
be compatible with organic compounds (particularly soaps).
• Is
not inactivated by living tissues.
• Is
non-corrosive
• Is
esthetically pleasing (non-staining or odorless)
Alcohols
and related compounds:-
• Antibacterial
action is due to their ability to denature the bacterial proteins & inhibit
phosphorylation systems
Ethanol
• Clear,
Colorless, Volatile liquid, Burning taste, Characteristic pleasant odor
• Antiseptic
• Preservative
• Mild counterirritant
• Rubbing
alcohol is used as an
ü Astringent
ü Rubefacient
ü Mild
local anesthetic
Isopropyl Alcohol
• Suitable
substitute for ethanol
• But
must not be ingested
• Primarily
as a disinfectant for the skin and for surgical instruments
• Bactericidal
in the concentration range of 50% to 95%
• A
40% concentration is considered equal in antiseptic efficacy to a 60% ethanol
in water solution
Formaldehyde Solution
• Formalin
is a colorless aqueous solution
• Used
as a disinfectant for surface sterilization
• Contains
not less than 37% w/v of formaldehyde (HCHO) with methanol added to retard
polymerization
• The
germicidal action of formaldehyde is slow but powerful
• The
mechanism of action: denaturation of proteins
ü Direct
nonspecific alkylation of nucleophilic functional group (amino, hydroxyl, and
sulfhydryl) in proteins and nucleic acids to form carbinol derivatives
• Gutaraldehyde
used to sterilize surgical instruments and surfaces contaminated with hepatitis
virus
Phenol
• Phenol
(carbolic acid) is a colorless to pale-pink crystalline material with a
characteristic “medicinal odor
• Liquified
phenol is simply phenol containing 10% water
• Bacteriostatic
conc- 0.5%, Bactericidal-1%, fungicidal 1.5%
• MOA:
denaturation of proteins
• Phenols
& derivatives:-
p-Chlorophenol
• p-Chlorophenol
is used in combination with camphor in liquid petrolatum as an external
antiseptic and anti-irritant
Hexachlorophene
• Hexachlorophene,
2,2-methylene bis (3,4,6-trichlorophenol)
• 2,2-
dihydroxy-3,5,6,3,5, 6-hexachlorodiphenylmethane
• Hexachlorophene
is easily adsorbed onto the skin and Enters the sebaceous glands
• Topical
application elicits a prolonged antiseptic effect
• Hexachlorophene
is used in concentrations of 2% to 3% in
• Soaps
• Detergent
creams
• Lotions
• Shampoos
• For
various antiseptic uses
• Effective
against gram-positive bacteria
• Many
gram-negative bacteria are resistant
Resorcinol
• m-
Dihydroxybenzene (resorcin)
• Resorcinol
is only a weak antisepticit is used in 1% to 3% solutions
• Ointments
and pastes in concentrations of 10% to 20% for the treatment of skin conditions
ü Ringworm
ü Eczema
ü Psoriasis
ü Seborrheic
ü Dermatitis
• Keratolytic agent
Hexylresorcinol
• 4-hexylresorcinol
• Effective
antiseptic
• Bactericidal
and fungicidal properties
Oxidizing agents:-
• Oxidizing
agents that are of any value as germicidal agents depend on their ability to
liberate oxygen in the tissues
• MOA:
oxidation of sulphydryl groups of bacterial enzymes
• Hydrogen
peroxide – H2O2
• Disinfectant
and sterilant
• Benzoyl
peroxide is both keratolytic and keratogenic
• It is used in the treatment of acne- exfoliant
,sebostatic
• Benzoyl peroxide induces proliferation of
epithelial cell leading to sloughing and repair
Halogen – containing compounds:
Iodine tincture
• It
is usually 2–7% elemental iodine, along with potassium iodide or
sodium iodide, dissolved in a mixture of ethanol and
water.
• It
contains iodine which is an antiseptic. It is for use on
minor wounds, cuts and scrapes.
Povidone–Iodine
• Charge-transfer
complex of iodine with the nonionic surfactant PVP (poly vinyl pyrrolidine)
• Povidone–iodine
is used as an aqueous solution for pre-surgical disinfection of the incision
site
• Treating infected wounds and damage to the skin
• It
is effective for local bacterial and fungal infections
Chlorine- Containing compounds
• All
the chlorophores act by releasing chlorine which oxidezes the sulphydryl groups
of bacterial enzymes and deactivates certain bacterial enzymes
• These
compounds release hypochlorous acid when dissolved in water in the presence of
acid – i.e HOCl is the active germicidal species.
• HOCl generates nascent oxygen to destroy the
vital cellular machinery of microorganism
Chlorhexidine
• Chlorhexidine,
also known as chlorhexidine gluconate (CHG), is a disinfectant and antiseptic
that is used for skin disinfection before surgery and to sterilize surgical
instruments-used both to disinfect skin of the patient and the hands of the
healthcare providers.
• It
is a germicidal mouthwash that reduces bacteria in the mouth. Chlorhexidine
gluconate oral rinse is used to treat gingivitis (swelling, redness, bleeding
gums).
Halazone
• P-dichlorosulfamoylbenzoic
acid
• Faint
chlorine odor
• The
sodium salt of halazone is used to disinfect drinking water
Cationic
surfactants:-
All cationic surfactants are quaternary ammonium compounds.
They are always ionized in water and exhibit surface active properties.
They form micelles by concentrating at the interface of
immiscible solvents-cationic head group has a high affinity for water and long
hydrocarbon tail has an affinity for lipids and nonpolar solvents
• Benzalkonium
chloride [Alkyl Benzyl dimethyl ammonium chloride is a mixture of alky benzyl
dimethyl ammonium chloride]
• Benzalkonium
chloride is a detergent, an emulsifier
and a wetting agent
• It
is used as an antiseptic for skin and mucous membranes
• Cetyl
pyridinium chloride
• It
is used as a general antiseptic for
intact skin
• Irrigation
of mucous membranes
Dyes
Gentian violet
[Hexamethyl-p-rosaniline chloride](crystal violet,methyl violet)
• Antiseptic
dye used to treat fungal infections of the skin (e.g., ringworm, athlete’s
foot).
• Also
has weak antibacterial effects and may be used on minor cuts and scrapes to
prevent infection.
Methylene blue
[3,7-Bis(dimethylamino)-Phenazathonium chloride
• Weak
antiseptic properties(bacteriostatic)
• Treatment
of cystitis & urethritis
Nitrogen Compound:
Nitrofuran derivatives
• Nitrofurazone:
[5-Nitro-2-furfuraldehyde semicarbazone]
• Furazolidone
{3-[(5-nitrofurylidene) amino]-2-oxazolidinone}
• Has
bactericidal action against a relatively broad range of intestinal pathogens
including S.aureus, E.coli, Salmonella, Shigella, Proteus, Enterobacter and
Vibriocholerae.
• Also
active against the protozoan Giardia Lamblia.
• Used
orally in the treatment of bacterial or protozoal diarrhea caused by
susceptible organisms.
Mercury compounds:
Mercurials
• From
early days, mercurial were used to treat skin infections & syphilis
• MOA:
reversible nature of sulfhydryl group blockage
• Nitromersol
[3-(hydroxy mercuri)-4-nitro-o-cresol]
• Thimerosal
{sodium [(o-carboxy phenyl) thio] ethyl mercury}
Mode of Action
Alcohols and related
compounds:-
• Ability
to denature proteins & inhibit phosphorylation
systems
• As
the primary alcohol chain length increases, vanderwaal’s interactions increase
and the ability to penetrate microbial membranes increases.
• As
water solubility decreases the apparent anti-microbial potency diminishes.
Branching of the alcohol chain decreases anti-bacterial potency. Weaker
vanderwall’s forces brought about by branching do not penetrate bacterial cell
membranes as efficiently. Yet, 2-propanol is used commercially instead of
n-propyl alcohol as it is less expensive.
• The
germicidal action of formaldehyde is slow but powerful. It is the direct,
non-specific alkylation of nucleophilic functional groups (amino, hydroxyl,
sulphydryl) in proteins and nucleophilic acids to form ‘Carbinol’ derivatives.
Phenols and
Derivatives:-
• Phenols “denatures bacterial proteins at low
concentrations”, “lysis of bacterial cell membranes at higher concentrations”
Oxidizing agents:-
• Germicidal
action is based on their ability to liberate oxygen in the tissues.
• All
these react in the tissues to generate oxygen and oxygen radicals. Oxidizing
agents are especially effective against anaerobic bacteria and can be used in
cleansing contaminated wounds.
Halogen-containing
compounds:-
• Iodine
acts to inactivate proteins by iodination of aromatic residues (Phenyl alanyl
and tyrosyl) and oxidation (sulphhydryl groups).
• Chlorine
released from compounds (Halozone) act by chlorination of amide nitrogen atoms
&oxidation of sulfhydryl groups in proteins
Cationic
Surfactants:-
• The
mechanism of action involves the dissolution of the surfactant into the
microbial cell membrane, destabilization and subsequent lysis.
• The
surfactants may also interfere with enzymes associated with the cell membrane.
Dyes:-
• Acts
on cell membranes. The difference in the susceptibility is related to the
cellular characteristics. The cationic dyes are active against gram +ve
bacteria and many fungi; gram –ve bacteria are generally resistant.
Nitrofurans:-
• The
mechanism of action of Nitrofurans are not fully understood.
• These
Nitrofurans are known to be mutagenic and carcinogenic under certain
conditions. The cellular effects may be due to DNA damage caused by metabolic
reaction products.
Preservatives
Ø “Preservatives
are added to various dosage forms and cosmetic preparations to prevent
microbial contamination”
Ø In
parenteral and ophthalmic preparations, preservatives are used to maintain
sterility in the event of accidental contaminations during use.
Ø Ideal
preservative should be
• Effective
at low concentrations against all possible microorganisms
• Non-toxic
• Compatible
with other constituents used in the
preparation
• Stable
for the shelf life of the preparation.
Ideal preservative does not exist – most preservatives have
some ideal features
• The
preservatives are of 2 types
Ø Para-hydroxy
benzoic acid derivatives
Ø Miscellaneous.
p-hydroxybenzoic acid
derivatives (Parabens):-
• Esters
of p-hydroxy benzoic acid have anti-fungal properties. Their toxicity to
human host is generally low due to rapid hydrolysis to p-hydroxy benzoic acid,
which is rapidly conjugated and excreted.
• Used
as preservatives for liquid dosage forms
• Preservative
effect increase with increase in molecular weight, but the methyl ester is more
effective against ‘Molds’, where as the propyl ester is more effective against
‘Yeasts’.
• Eg,
Methyl paraben – Methyl-p-hydroxybenzoate.
Others are propyl paraben (Propyl-p-hydroxybenzoate), Ethyl
paraben (Ethyl-p-hydroxybenzoate) and Butyl paraben (Butyl-p-hydroxybenzoate)
Other preservatives:-
• Chlorobutanol:
– 1,1,1-Trichloro-2-Methyl-2-Propanol
• Used
as bacteriostatic in injections, ophthalmics & intranasal preparations
• Sodium
benzoate-preservative in acidic liquid preparations
• Phenyl
carbinol- used in ointments & lotions as an antiseptic in the treatment of
pruritic infections
• Sodium
propionate, Benzyl alcohol, Phenyl ethyl alcohol (2-Phenyl ethanol), Benzoic
acid, Phenyl mercuric nitrate, Phenyl mercuric acetate, etc