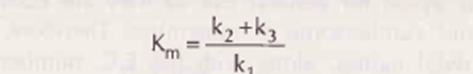Factors effecting enzyme activity

Objective
At the end of this lecture, student will be able to
• Explain the factors affecting enzyme activity
• Describe enzyme kinetics
• Outline Michealis-Menton Equation
Factors effecting enzyme activity
• The important factors that influence the velocity of the enzyme reaction are
1. Concentration of substrate
2. Concentration of enzyme
3. Temperature
4. PH
5. Effect of product concentration
6. Effect of activators
7. Effect of time
8. Effect of light and radiation
1. Concentration of substrate
• Increase in the substrate concentration gradually increases the velocity of enzyme reaction within the limited range of substrate levels
• A rectangular hyperbola curve is obtained when velocity is plotted against the substrate concentration
• 3 distinct phases of the reaction are observed in the graph;
A-linear; B-curve; C-almost unchanged
Michelis-Menten Equation:
Order of reaction:
• When the velocity of the reaction is proportional to the substrate concentration (i.e [S] ˂ KM), the rate of the reaction is said to be first order with respect to substrate
• When the [S] ˃ KM, the rate of reaction is independent of substrate concentration and the reaction is said to be zero order
• Enzyme kinetics and K value: The enzyme (E) and substrate (S) combine to form an unstable enzyme-substrate complex (ES) and product (P)
• Here kl, k2 & k3 represent the velocity constants for the respective reactions as indicated by arrows
• KM Michaelis-Menten constant (or Brigrs and Haldane’s constant) is given by the formula
• The following equation is obtained after suitable algebraic manipulation
• Let us assume that the measured velocity (v) is equal to ½ Vmax. Then the equation (1) may be substituted as follows;
• K stands for a constant and m stands for Michaelis (in KM)
• KM or Michaelis-Menten constant is defined as the substrate concentration (expressed in moles/l) to produce half-maximum velocity in an enzyme catalysed reaction.
• KM value is a constant and a characteristic feature of a given enzyme. lt is representative for measuring the strength of ES complex
• A low KM value indicates a strong affinity between enzyme and substrate, whereas a high KM value reflects a weak affinity between them
• For majority of enzymes, the KM values are in the range of 10-5 to 10-2 moles
Line weaver-Burk double reciprocal plot:
• For the determination of KM value, the substrate saturation curve is not very accurate, Since Vmax is approached asymptotically
• By taking the reciprocals of the equation (1) a straight line graphical representation is obtained
• The above equation is similar to y = ax + b
• Therefore, a plot of the reciprocal of the velocity [1/v] VS reciprocal of the substrate concentration [1/[s]] gives a straight line
• Here the slope is KM/Vmax and whose y intercept is 1/Vmax
• By this plot, it is much easier to calculate the KM from the intercept on x-axis which is -(1/Km)
• Further, the double reciprocal plot is useful in understanding the effect of various inhibitions
2. Concentration of enzyme
• Increase in concentration of the enzyme, increased the velocity of the reaction
• This property of enzyme is used to determining the serum enzymes for the diagnosis of diseases
• By using a known volume of serum and keeping all the other factors (substrate, pH, temperature etc.) at the optimum level, the enzyme could be assayed in the laboratory
3. Temperature:
• Velocity of an enzyme reaction increases with increase in temperature up to a maximum and then declines
• A bell-shaped curve is usually observed
• Temperature coefficient or Q10 is defined as increase in enzyme velocity when the temperature is increased by 10oC
• For a majority of enzymes, Q10 is between 0oC and 40oC
• The optimum temperature for most of the enzymes is between 40oC- 45oC
• Few enzymes (e.g. venom phosphokinases) are active even at 100oC
• Some plant enzymes like urease have optimum activity around 60oC
• In general, when the enzymes are exposed to a temperature above 50oC Denaturation occurs and the enzymes become inactive
4. pH
• Increase in the hydrogen ion concentration (pH) considerably influences the enzyme activity and a bell-shaped curve is normally obtained
• Each enzyme has an optimum pH at which the velocity is maximum, Below and above this pH, the enzyme becomes totally inactive
• Most of the enzymes show optimum activity around neutral pH (6-8)
• However some enzymes like pepsin at (1-2), acid phosphatase (4-5) and alkaline phosphatase (10-11) are active
5. Effect of product concentration
• The accumulation of reaction products generally decreases the enzyme velocity
• Here, the products combine with the active site of enzyme and form a loose complex and thus inhibit the enzyme activity
• But in the living system, this type of inhibition is generally prevented by a quick removal of products formed
6. Effect of activators
• Some of the enzymes require certain inorganic metallic cations like Mg2+, Mn2+,zn2+, ca2+, co2+, cu2+, Na+, K+ etc for their optimum aciivity
• Rarely, anions are also needed for enzyme activity e.g. chloride ion amylase
• Metals function as activators of enzyme velocity through various mechanisms
Combining with the substrate
Formation of ES-metal complex
Direct participation in the reaction
Bringing a conformational change in the enzyme
• Two categories of enzymes requiring metals for their activity are distinguished
Metal activated enzymes: here metal is not tightly held by the enzyme and can be exchanged easily with other ions e.g. ATPase (Mg2+ and Ca2+) Enolase (Mg2+)
Metalloenzymes: These enzymes hold the metals rather tightly which are not readily exchanged. e.g. alcohol dehydrogenase, carbonic anhydrase, alkaline phosphatase, carboxypeptidase and aldolase contain zinc
Phenol oxidase (copper), Pyruvate oxidase (manganese), Xanthine oxidse (molybdenum), Cytochrome oxidase (Iron and copper)
7. Effect of time:
• Under ideal and optimal conditions, the time required for an enzyme reaction is less. Variations in the time of the reaction are generally related to the alterations in pH and temperature
8. Effect of light and radiation
• Exposure of enzymes to ultraviolet, beta, gamma and X-rays inactivates certain enzymes due to the formation of peroxides. E.g. UV rays inhibit salivary amylase activity
Summary
• Increase in the substrate concentration gradually increases the velocity of enzyme reaction within the limited range of substrate levels
• Increase in concentration of the enzyme, increased the velocity of the reaction
• Velocity of an enzyme reaction increases with increase in temperature and pH up to a maximum and then declines
• The accumulation of reaction products generally decreases the enzyme velocity
• Exposure of enzymes to ultraviolet, beta, gamma and X-rays inactivates certain enzymes
Also, Visit:
B. Pharma Notes | B. Pharma Notes | Study material Bachelor of Pharmacy pdf