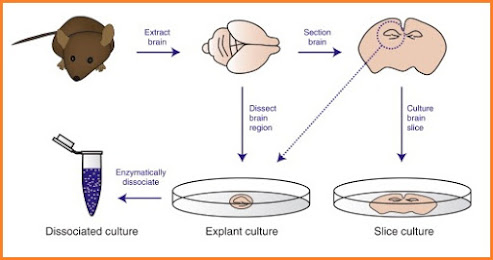
Cell Culture

Cell culture refers to the removal of cells from an animal or plant and their subsequent growth in a favorable artificial environment.
The cells may be removed from the tissue directly and disaggregated by enzymatic or mechanical means before cultivation, or they may be derived from a cell line or cell strain that has already been established.
It is widely used for growing viruses.
Tissues are dissociated into the component cells by the action of enzyme and mechanical shaking.
The cells are washed, counted and suspended in a growth medium.
The growth medium conists of essential amino acids, glucose, vitamins, salts and a buffer. Antibiotics are added to prevent bacterial contamination.
The cell suspension is put into bottles, tubes and pertidishes.
The cell adhere to the glass or plastics surface, divide and form a confluent monplayer sheet within a week.
Cell culture is further classified based on origin,chromosomal characters and the number of generations through which they can be maintained .
Types of Cell Culture
1. Primary cell culture
2. Diploid cell strain
3. Continuous cell culture
Primary cell culture
They are normal cells freshly taken from the body and cultured.
They are capable of only limited growth in culture.
They cannot be maintained in serial culture example: – monkey kidney, human embryo kidnry and chick embryo cell culture.
Primary cultures are derived directly from excised, normal animal tissue and cultures either as an explant culture or following dissociation into a single cell suspension by enzyme digestion.
Such cultures are initially heterogeneous but later become dominated by fibroblasts.
The preparation of primary cultures is labour intensive and they can be maintained in vitro only for a limited period.
During their relatively limited lifespan primary cells usually retain many of the differentiated characteristics of the cell in vivo.
Primary cultures by definition have not been passaged, as soon as they are passaged they become a cell line and are no longer primary. ‘Primary’ cells sourced from most suppliers are in fact low- passage cell lines.
Diploid cell
Diploid cell strains are cells of a single type that retain the original diploid chromosome number and karyotype during serial subcultivations for a limited number of times .
After about fifteen serial passages, they undergo ‘senescence’.
Diploid strain developed from human fibroblast are a good example .
Continuous cell lines
These are cells of a single type, usually derived from cancer cells. they are capable of continuous serial cultivation indefinitely.
Hela cell are derived from carcinoma of cervix. Cell culture is used for the isolation of viruses an their cultivation for vaccines production.
Continuous cultures are comprised of a single cell type that can be serially propagated in culture either for a limited number of cell divisions (approximately thirty) or otherwise indefinitely.
Cell lines of a finite life are usually diploid and maintain some degree of differentiation.
The fact that such cell lines senesce after approximately thirty cycles of division means it is essential to establish a system of Master and Working banks to maintain such lines for long periods.
Continuous cell lines that can be propagated indefinitely generally have this ability because they have been transformed into tumour cells.
Tumour cell lines are often derived from actual clinical tumours, but transformation may also be induced using viral oncogenes or by chemical treatments.
Transformed cell lines present the advantage of almost limitless availability, but the disadvantage of having retained very little of the original in vivo characteristics.
Cell Line
After the first subculture, the primary culture becomes known as a cell line or subclone.
Cell lines derived from primary cultures have a limited life span (i.e., they are finite; see below), and as they are passaged, cells with the highest growth capacity predominate, resulting in a degree of genotypic and phenotypic uniformity in the population.
cell strain
If a subpopulation of a cell line is positively selected from the culture by cloning or some other method, this cell line becomes a cell strain.
A cell strain often acquires additional genetic changes subsequent to the initiation of the parent line.
Procedure for cell culture:-
1. Incubation temperature should be 36°C.
2. The pH for growth should be 7.2 and 7.4.
3. The level of glucose and L-glutamine influence the growth of cell.
4. The range of inorganic ions and vitamins are essential for cell survival.
5. Both oxygen and carbon dioxide are essential and are provided either as a mixture of Co2 and air supplied to the culture vessel or by sealing the vessel tightly to retain the Co2 Produced by cell metabolism.
6. Isolation of cells – cells isolated from tissue in in-vivo (in a test tube) either by enzymatic digestion with enzymes such as collagenase, trypsin or protease or Pieces of tissue can be placed in growth tissue and the cells that grow out are available for culture This method is known as explant culture can be used.
7. Maintaining cells in culture viable (alive). Providing essential nutrients in the media is important (Amino acids, carbohydrates, vitamins, minerals, growth hormones and essential gases o2, co2).
8. Maintaining Aseptic technique- sterilize all glassware for handling cell culture and media.
Technique for Primary Culture – Preparation of primary culture
Three techniques are most commonly used:-
1. Mechanical Disaggregation-
2. Enzymatic Disaggregation
3. Primary Explant Technique- developed by Harrison in 1907
Applications of Animal Cell Culture
1. Provide a model system for basic cell biology and biochemistry.
2. Toxicity Testing.
3. Cancer Research.
4. Virology
5. Cell-based manufacturing
6. Genetic Counselling.
7. Genetic Engineering.
8. Drug Screening and Development
9. Gene Therapy.