Formulation of
suspensions
Learning
Objectives
• At the end of this
lecture, student will be able to
• Explain the
formulation of suspensions
• List out
methods of dispensing suspensions
• Give examples
of suspensions
Formulation
of Suspensions
1. Medicament/Drug
2. Flocculating agents
3. Deflocculating agents
4. Suspending agents
5. Thickening agents
6. Wetting agents
7. Preservatives
8. Organoleptic additives
1.
Flocculating agents
– induce flocculation
E.g
a) Electrolytes: sodium salts of acetates, phosphates and
citrates.
b) Surfactants: ionic and non-ionic
c) Polymers – starch, alginates, cellulose derivatives,
Tragacanth, carbomers and silicates
2. Deflocculating
agents
– induce deflocculation
E.g Ionic surfactants
Ionic surfactants as flocculating and deflocculating agents
3. Suspending agents
– Reduce the rate of sedimentation of the dispersed
particles.
– Increasing the viscosity of the external phase
E.g.
i) Tragacanth BP (internal or external suspensions)
ii) Compound Tragacanth Powder BP (containing: 15%
Tragacanth BP, 20% Acacia BP, 20% Starch BP and 45% Sucrose BP)
iii) Bentonite BP (external suspensions)
4. Thickening agents
– Increase the
viscosity of the continuous phase
– Particles remain suspended for a sufficiently long time
– Easy to withdraw a uniform dose
– Viscosity of the preparation –easy pouring from the
container and transferring to the site of application
E.g.
1. Natural polysaccharides-
Acacia, Tragacanth, Alginates
2. Semi synthetic (cellulose derivatives)- Methyl cellulose,
SCMC
3. Inorganic agents – Bentonite, Magnesium aluminium
silicate (Veegum)
4. Synthetic
compounds – Carbomers, Colloidal silicon dioxide (Aerosil)
5. Wetting agents
– Reduce the interfacial tension between the solid particles
and the liquid medium
– Increases the affinity of the particles towards the
surrounding medium
– Helps in penetration of liquid into the particles
– This produces a good suspension. E.g. Spans and Tweens,
Saponins
6. Preservatives
– To protect the suspension against bacterial growth
– Should be effective against a wide range of microorganisms
– Should be chemically & physically compatible with the
other ingredients
E.g. Benzoic acid, Parabens (methyl and propyl paraben)
7. Organoleptic
additives
– Flavouring agents: for a distinct flavour
E.g. Vanilla flavour, banana flavour, strawberry.
– Sweetening agents: to provide sweetness
E.g. Sucrose, Saccharin sodium, aspartame
– Colouring agents: to increase aesthetic appeal
E.g. Amaranth, tartrazine
– Perfumes: to increase patient acceptability
E.g. Rose water, Lavender oil.
Methods of Dispensing Suspensions
1) Suspensions containing Diffusible solids
2) Suspensions containing Indiffusible solids
3) Suspensions containing precipitate forming liquids
4) Suspensions containing poorly wettable solids
5) Suspensions produced by chemical reaction
1) Suspensions
containing Diffusible solids
Preparation:
a) The drug is finely powdered with the other solid
ingredients.
b) 3/4th of the vehicle is added to the powder mixture and
triturated to form a smooth cream.
c) More vehicle is added.
d) The suspension is filtered to remove any foreign
particles.
e) Any other liquid ingredients if present, is added and the
suspension is made upto volume.
f) The suspension is transferred to a bottle & shaken
well.
E.g. Rx Light Kaolin
Light magnesium carbonate Sodium bicarbonate Peppermint
water
Labeling: SHAKE WELL BEFORE USE
2) Suspensions
containing Indiffusible solids
• They contain a suspending agent / thickening agent
• Delay the sedimentation rate & prevent the formation
of aggregates.
• The thickening agents used are:
i) Compound Tragacanth powder: mixture of powdered
tragacanth, acacia, starch & sucrose. It is used when the vehicle is not
water or chloroform water.
ii) Tragacanth mucilage: mixture of tragacanth powder, 90%
ethanol & chloroform water.
•It is used only when the vehicle is water or chloroform
water
• If it is added to preparations containing medicinally
active vehicle, it may replace the medicinally active vehicle, thereby
decreasing its activity.
3) Suspensions
containing precipitate forming liquids
• Liquid preparations containing resinous matter when mixed
with water show precipitation of resin
• It sticks to the sides of the bottle & will not
diffuse on shaking.
E.g. Compound benzoin tincture, Tolu tincture
• To prevent this, suspending agents like Compound
Tragacanth powder or tragacanth mucilage is added.
4) Suspensions
containing poorly wettable solids
• Substances like sulphur & hydrocortisone – are
insoluble in water & poorly wetted by it
• In simple aqueous dispersions, it is difficult to disperse
clumps
• The foam produced on shaking is slow to subside since it
is stabilized by the film of unwettable solid at the liquid/air interface.
•To ensure wetting, the interfacial energy between the solid
& liquid must be reduced.
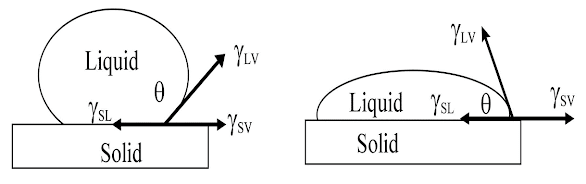
Wetting is
measured by determining the contact angle of a liquid or solid
High contact angle = poor wetting Low contact angle = good wetting
• Wetting is achieved by adding a suitable wetting agent
• This is adsorbed at the solid liquid interface
• The affinity of the particles for the surrounding medium
is thus increased
E.g. Sulphur lotion – Quillaia tincture is used as the
wetting agent. The saponins in quillaia extract have been used to suspend
sulphur in lotion.
5) Suspensions
produced by chemical reaction
• The insoluble active constituent of a lotion is produced
by a chemical reaction
• Finer precipitate –
if dilute solutions of the reactants are mixed
• Hence the reacting
substance should be
dissolved separately in approx. half volumes of the vehicle and the 2
parts mixed.
• The precipitate is diffusible & no suspending agent is
required.
E.g 1. Magnesium Hydroxide mixture
2. Zinc Sulphide lotion B.P.C (used to treat acne &
scabies)
Rx Sulphurated
potash
Zinc
sulphate
Concentrated
camphor water
Water
• Sulphurated potash is a mixture of potassium polysulphides
& other sulphur containing compounds
• Should be freshly prepared as its solubility decreases on
storage
• If sulphurated potash is added to the zinc sulphate: the
ppt. is diffusible
• If zinc sulphate is added to sulphurated potash: the ppt.
is indiffusible.
• Protection from light is necessary to reduce oxidation of
the Sulphide to sulphite or sulphate.
Summary
1. Formulation of
Suspensions:
– Medicament/Drug: Insoluble drug
– Flocculating agents: Induces flocculation
– Deflocculating agents: Induces deflocculation
– Suspending agents: Delay sedimentation rate
– Thickening agents: Increases viscosity to delay sedimentation
rate
– Wetting agents: Helps in penetration of liquid into the particles
– Preservatives: Prevent microbial contamination
– Organoleptic additives: Imparts sweetness, increases
palatibility and aesthetic appeal
2. Methods of
dispensing
– Diffusible suspensions
– Indiffusible suspensions
– Suspensions containing precipitate forming liquids
– Suspensions containing poorly wettable solids
– Suspensions produced by chemical reaction