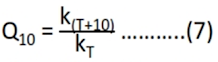Drug Stability

Learning Objectives
At the end of this lecture, students will be able to
- Describe the physical factors affecting the degradation of pharmaceutical products
- Describe the chemical factors causing the degradation of drugs and the preventive measures
- Describe the influence of light and temperature on drug Degradation
- Describe the accelerated stability testing of drug products
- Explain the different methods of conducting accelerated stability testing
Drug Stability
- Drug decomposition or degradation occurs during storage due to chemical alteration of the active ingredients
- Stability is defined as the time lapse during which the drug product retains the same properties and characteristics that it possessed at the time of manufacture
- Stability studies are necessary for the following reasons:
– Chemical degradation may lead to under medication
– Decomposition of active drug may lead to formation of toxic product
– Instability may be due to change in physical
Drug stability- Physical Degradation of Pharmaceutical Product
- The different forms of physical degradation of pharmaceutical products are:
– Loss of volatile constituents
– Loss of water
– Absorption of water
– Crystal growth
– Polymorphism
– Colour change
Drug Stability- Chemical Decomposition and Preventive Measures
Hydrolysis
- The principles that governs the hydrolysis reactions are:
– Drugs with ester and amide groups
– Drugs in the form of weak acids or weak bases
– Hydrolysis reaction are catalysed by H+ and (OH)-
- Drugs which decompose by hydrolytic pathways are: Aspirin, Atropine, Chloramphenicol, Cephalosporins etc.
Protection against hydrolysis
– Buffers
– Complexation
– Suppression of solubility
– Removal of water
Oxidation
- The general principles that govern oxidation reaction are:
– Presence of atmospheric oxygen
– Light energy induced free radical chain reaction
– Presence of trace metals
– Organic peroxide induced chain initiation
– Drugs in the form of weak acids or weak bases
– Oxidation reaction catalyzed by H+ and OH-
- Drugs which decompose by oxidation reaction are: Promethazine, Epinephrine, Vitamin A, Riboflavin, ascorbic acid etc.
Protection against oxidation
– Antioxidants
– Chelating agents
– Vehicles
– Micellar solubilisation
– Buffers
Influence of Light on Drug Stability
- Light energy activate molecules and enhance the rate of a reaction
- Drugs which undergo light induced chemical degradation are called photolabile (photosensitive) drugs
- Examples of photosensitive drugs are Riboflavin, Tetracycline, Chlorpromazine etc.
- The photochemical decomposition of pharmaceuticals are due to absorption of sunlight in the spectral region of visible blue and violet U.V wavelengths (500- 300 nm)
Influence of Temperature on Drug Decomposition- Arrhenius Equation
- The speed of many reactions increases two to three times with every 100 C rise in temperature
- Arrhenius equation explains the effect of temperature on the rate of a reaction
Where, k= specific rate constant
A= Arrhenius or frequency factor
Ea= energy of activation
R= ideal gas constant
T= absolute temperature
• Taking logarithms on both side of equation (1)
Converting equation (2) to log base 10 gives
• Energy of activation is the minimum energy a molecule should possess so that the molecular collision produce the product
• Arrhenius factor is defined as the frequency of collisions which can occur between the molecules
Chemical Stability Testing-Accelerated Stability Studies
• The method of accelerated testing of pharmaceutical products based on the principles of chemical kinetics
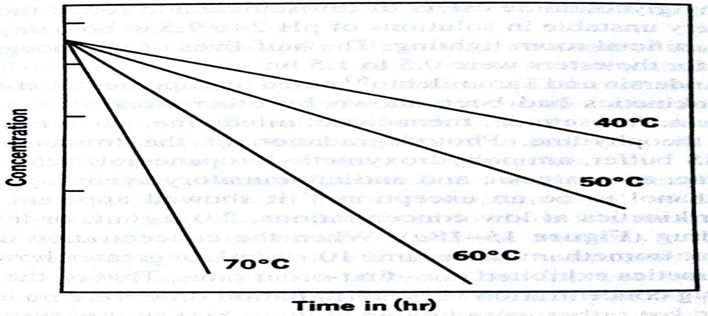
Method 1
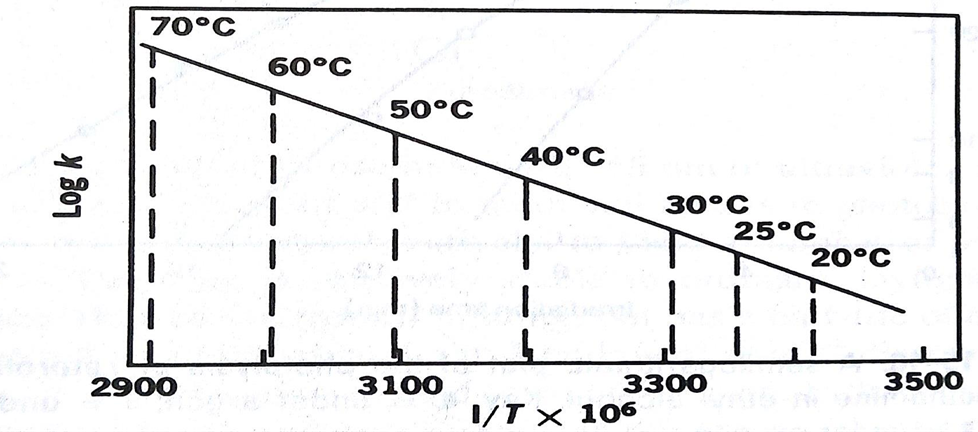
• The k values for the decomposition of a drug in solution at various elevated temperature are obtained by plotting some function of concentration against time
• The logarithms of the specific rates of decomposition are then plotted against the reciprocals of the absolute temperature
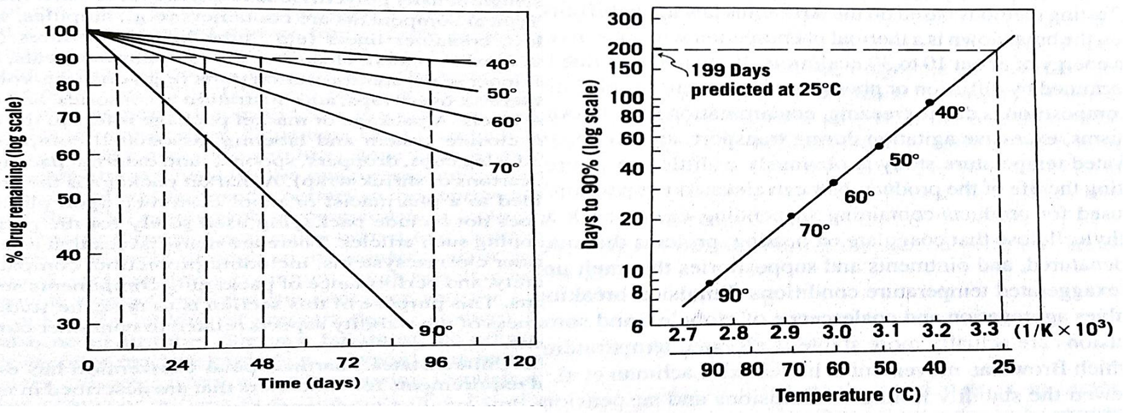
Method 2
• Fractional life period is plotted against reciprocal temperatures and the time in days required for the drug to decompose at room temperature is obtained
Method 3- Nonisothermal kinetics
• Temperature and time are related through an appropriate function:
1/T = 1/T0 + at……………..(4)
Where T0 is the initial temperature and a is a reciprocal heating rate constant
• The Arrhenius equation for time zero and time t can be written as
• Substituting equation (4) into (5) gives:
• Because temperature is a function of time, t, a measure of stability kt, is directly obtained over a range of temperatures
Method 4- Stress testing
• The testing typically includes the effects of temperature in 100 C increments above that for accelerated testing
• Other conditions like humidity (75% relative humidity and greater), where appropriate oxidation, photolysis are also considered
• This testing is undertaken to assess the effect of severe conditions on the drug product
Method 5 – Q10 method
• Using this method, the effect of 100 C rise in temperature on the stability of pharmaceuticals can be estimated
• Q10 can be defined by the equation:
• Q10 can be calculated from the equation:
Methods of Conducting Accelerated Stability Testing
Several methods are used for accelerated stability testing:
Arrhenius Equation
This equation relates the rate of a chemical reaction to temperature, allowing the prediction of degradation rates at different temperatures.
High-Temperature Stability Testing
Samples are exposed to elevated temperatures for a defined period to observe any changes in physical or chemical properties.
Cyclic Temperature Testing
Samples are subjected to alternating cycles of high and low temperatures to mimic real-world temperature fluctuations.
Humidity Testing
Testing at elevated humidity levels helps assess the impact of moisture on drug stability.
Photostability Testing
Exposing samples to UV and visible light helps assess the effects of light exposure on drug degradation.
FAQs
Why is drug stability important? Drug stability ensures that pharmaceutical products retain their effectiveness and safety over time, preventing degradation-related issues.
What are common preventive measures against drug degradation? Preventive measures include proper packaging, storing in controlled environments, and the addition of stabilizing agents like antioxidants.
What is the purpose of accelerated stability testing? Accelerated stability testing provides a faster assessment of how drugs degrade under extreme conditions, helping to predict long-term stability.
Can temperature fluctuations affect drug stability? Yes, temperature fluctuations can accelerate chemical reactions and lead to drug degradation. Storing drugs in controlled environments is vital.
What is photostability testing? Photostability testing involves exposing drug samples to light, especially UV light, to assess their susceptibility to photodegradation.
Summary
• The different forms of physical degradation of pharmaceutical products are: Loss of volatile constituents, Loss of water, Absorption of water, Crystal growth, and polymorphism
• Chemical factors affecting the degradation of drugs are hydrolysis and oxidation
• Light energy activate molecules and enhance the rate of a reaction
• The speed of many reactions increases two to three times with every 100 C rise in temperature
• Arrhenius equation explains the effect of temperature on the rate of a reaction
• Accelerate dstability studies can be conducted by:
– Graphical methods
– Nonisothermal kinetics
– Stress testing method
– Q10 calculation method
– Freeze thaw method
Also, Visit: Biotechnology Notes