Chemistry of Diphenylmethane
Chemistry
of Diphenylmethane
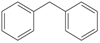
Diphenylmethane is an organic compound with the formula (C6H5)2CH2
or C13H12
The compound consists of methane wherein two hydrogen atoms
are replaced by two phenyl groups.
Diphenylmethane forms a common skeleton in organic
chemistry; the diphenylmethyl group is also known as benzhydryl.
Natural Occurring Sources –
1,1′-Methylenebisbenzene has been identified as a component in crude
oils
Synthesis
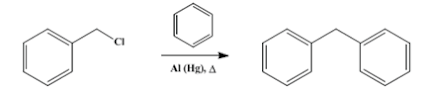
• The Friedel-Craft reaction of benzyl
chloride with benzene in the presence of aluminum chloride is an important
method for production of diphenylmethane.
• Other known catalysts of this process are
elemental aluminum, iron(III) chloride, antimony pentachloride, zinc
chloride-hydrogen chloride and boron fluoride dihydrate.
• When sulfuric acid is used as a
condensation agent diphenylmethane yields are 83% and with polyphosphoric acid
yields are over 90%.
• Condensation of benzene with formaldehyde in
presence of 85% sulfuric acid yields 79% diphenylmethane.
benzyl chloride diphenylmethane
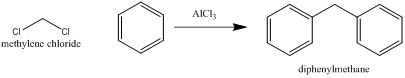
• Prepared
from methylene chloride and benzene with aluminum chloride as catalyst.
Properties of
Diphenylmethane:
Molecular Weight:
168.239 g/mol
Melting Point – 25.4 deg C
Odor of oranges
Solubility –
Freely soluble in alcohol, ether, chloroform, hexane, benzene; insoluble in
liquid ammonia
Summary
• Diphenylmethane
is an organic compound with the formula (C6H5)2CH2
or C13H12
• 1,1′-Methylenebisbenzene
has been identified as a component in crude oils
• Synthesised through Friedel-Craft
reaction
For PDF Notes Click on Download Button