Adverse
Drug Reaction Management
Content
• Identifying
adverse events
• Assessing
adverse events
• Reporting
adverse reactions
Objective
After completion of this lecture, student will be able
to:
• Identify
adverse events
• Assess
adverse events
• Report
adverse reactions
INTRODUCTION
Ø It
describes the procedure for identifying,
recording and reporting of Adverse
Reactions(AE) and Serious Adverse Reactions (SARs)
Ø It
also describes the procedure for reporting
Suspected Unexpected Serious Adverse Reactions(SUSARs)
ABBREVATIONS
Ø AE/AR Adverse Event/Reaction
Ø SAE/SAR Serious Adverse Event/Reaction
Ø SUSAR Suspected Unexpected Serious Adverse
Reactions
Ø CTIMP Clinical Trail of an Investigational Manufacturing
Product
Ø NIMP Non Investigational Medicinal Product
DEFINITIONS
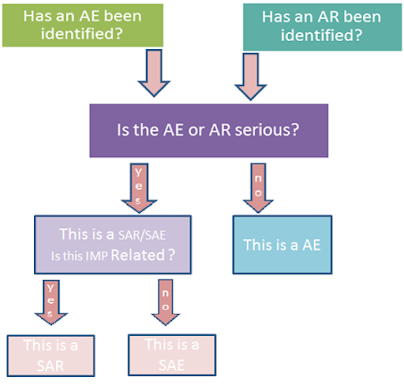
ADVERSE EVENTS
Any Untowarded medical occurance in a patient or a clinical trial
participant administered an investigational product, and which does not
necessarily have a causal relationship with the drug
ADVERSE REACTIONS
All untowarded and unintended responses to the IMP related
to any dose administered to that participant.
SERIOUS ADVERSE REACTIONS
Any untowarded
medical occurance that at any dose
Ø Results
in death
Ø Is
life-threatening
Ø Requires
hospitalisation,or prolongation of existing hospitalisation
Ø Results
in persistent or significant disability or incapacity
Ø Is
a congenital anomaly.
SUSPECTED
UNEXPECTED SERIOUS ADVERSE REACTIONS
• The
trail protocol or IB should include a list of known side effects for each drug
in the study.
• This should be consulted when a SAR occurs, to
determine expectedness. If the event is not listed, or has occurred in a more
serious form,or more frequently than expected, it should be considered to be a
SUSAR
Identifying of Adverse Events
• The
protocol should state whether any NIMPs are to be supplied to participants in
the trail.
• The
procedure for notifying such adverse events to the Principal Investigator must
be clearly documented in the trail protocol.
• The
trial protocol should define how AEs will be recorded, and states that all SAEs
will be reported to the Sponsor.
• AEs
may also be identified by support documents, for example clinical biochemistry,
hematology, and radiology.
Assessment of Adverse
Event
• AEs
must be assessed for seriousness, causality,
expectedness, and severity
• It
is the responsibility of the PI to assess each AE for seriousness, causality, expectedness, and
severity
• The
sponsor should review if a SAR reported to them is expected or not. If the
event is not expected it will be considered as a SUSAR.
Assessment of
Severity
Ø The
CI or PI or delegated medically qualified research team member should make an
assessment of severity for each AR.
Ø The
assessment should be recorded on the SAE form and reported to the sponsor
according to the following categories:
• Mild: A reaction that is easily tolerated by
the subject.
• Moderate: A reaction that is sufficiently
discomforting to interfere with normal everyday activities.
• Severe:
A reaction that prevents normal everyday activities
Reporting of Adverse reactions
Ø Patient
details
Initials
Gender
Age and
date of birth
Weight
Height
Ø Suspected
drugs
Generic
name of the drug *
Indication(s)
for which suspect drug was prescribed or tested.
Dosage
form and strength
Daily
dose and regimen (specify units – e.g., mg, ml, mg/kg) ..
Route of
administration.
Starting
date and time of day.
Stopping date and time, or duration of
treatment
Ø Other
Treatment(s)
Provide the same information for
concomitant drugs
Ø Details
of Suspected Adverse Drug Reaction(s) Full description of reaction(s) including
body site and severity, as well as the criteria for regarding the report as
serious,whenever possible, describe a specific diagnosis for the reaction
• Start
date (and time) of onset of reaction
• Stop
date (and time) or duration of reaction
Ø Outcome
Information
on recovery; results of specific tests and/or treatment.
For a
fatal outcome, cause of death and its possible relationship to the suspected
reaction; any post-mortem findings.
Any
Other information relevant to facilitate assessment of the case, such as
medical history of allergy, drug or alcohol abuse; family history; findings
from special investigations etc
Ø Details
about the Investigator*
Name
Address
Telephone
number
Profession
(speciality)
Date of
reporting the event to Licensing Authority:
Date of
reporting the event to Ethics Committee overseeing the site:
Signature
of the Investigator
Responsibilities
Responsibilities of
Sponsor
• SAE
should be reported to the licensing authority within 14 calendar days
• Submit
status report to the licensing authority
periodically
Responsibilities of Investigator
- Ensure
adequate medical care is provided to the subject
• SAE
and unexpected AE should be reported to
the sponsor within 24 hrs
• To the EC within 7 working days
• Regular
reporting of adverse events is done through data collection in case report
forms
• Adverse
events are already known as potential risks when participating in a research
study with a specific investigational product
• It
is important to have a documented baseline assessment that is detailed with
health history and current health issues prior to exposure to the
investigational product
• As
the participant is exposed to the investigational product, the research team
must document any changes in health that may be due to the research study
product and report these adverse events through normal data collection tools
provided by the sponsor
• Most
reports require a brief history of the event
• Key
information that is helpful to the reviewers of the reports are:
• participant
demographic information, when the participant started the research study, what
research intervention the participant was receiving,
• previous
medical history, concomitant medications
• last
exposure to investigational product,
• abnormal labs or diagnostic tests,
• when the study team was notified of the event,
description of the actual event, relationship of the AE to the investigational
product,
• whether the participant was hospitalized,
• the event has resolved or not,
• whether
the participant will be removed from the research study or continue to receive
the research product
IDENTIFYING
OF ADVERSE REACTIONS
Summary
• Adverse event – Any Untowarded medical
occurance in a patient or a clinical trial participant administered an
investigational product , and which does
not necessarily have a causal relationship with the drug
• Adverse reactions- All untowarded and
unintended responses to the IMP related to any dose administered to that
participant