Post Views: 84
GRAM STAIN TECHNIQUE
1. The Gram stain procedure was originally developed by the Danish physician Hans Christian Gram to differentiate “pneum ococci” from “Klebsiella pneum onia”.
2. In brief, the procedure involves the application of a solution of iodine (potassium iodide) to cells previously stained with crystal violet or gentian violet.
3. This procedure produces “purple colored iodine-dye complexes” in the cytoplasm of bacteria. The cells that are previously stained with crystal violet and iodine are next treated with a decolorizing agent such as 95% ethanol or a mixture of acetone and alcohol.
4. The difference between Gram-positive and Gram-negative bacteria is in the permeability of th e cell wall to these “purple colored iodine-dye complexes” when treated with the decolorizing solvent.
5. While Gram-positive bacteria retain purple iodine-dye complexes after the treatment with the decolorizing agent, Gram-negative (-ve) bacteria do not retain complexes when decolorized
6. To visualize decolorized Gram-negative bacteria, a red counter stain such as safranin is used after decolorization treatment
7. Appearance of the Gram positive “coccus” and Gram negative “bacillus” at different stages of the gram staining procedure are illustrated below:

Preparation of the smear
The first consideration is the correct preparation of the smear.
Make a thin film of the material on a clean glass slide, using a sterile loop or swab for viscous specimens.
Air dry, then heat fix the slide by passing it several times through a flame (the slide should not become too hot to touch).
Precaution-“Failure to follow these directions may cause staining artifacts and disrupt the normal morpholog y of bacteria and cells.”
To be visible on a slide, organisms that stain by the Gram method must be present in concentrations of a minimum of 104 to 105 organisms/ml of unconcentrated staining fluid.
At lower concentrations, the Gram stain of a clinical specimen seldom reveals organisms even if the culture is positive.
Smears that are not properly fixed tend to be washed away during staining and washing resulting in the absence of stained bacteria.
In special situations, the following guidelines may be helpful:
When cerebrospinal fluid (CSF) contains only a few organisms, they are more likely to be found if a concentrated “thick smear” is examined.
To prepare a “thick smear” the specimen is spun at high speed and a large drop of sediment (or multiple drops, drying in between each drop) is placed in the center of the slide and allowed to air dry.
The cytocentrifuge ma y prove to be useful in concentrating bacteria as well as in preserving cell morphology.
When fluid specimens such as urine or CSF seem to vanish after the staining procedure, a wax mark, placed near the smeared area on the slide (same side) after the staining procedure (to avoid introducing wax artifacts) will reduce frustration in locating the specimen under the microscope.
The wax mark can be used for quick focussing.
In a grossly bloody specimen, it may prove difficult to distinguish microorganisms from artifacts.
After air-drying and heat-fixing this type of specimen, the added preparatory step of covering it with distilled water, waiting five minutes, and then rinsing, may cause the red blood cells to lyse and float off.

Staining procedure
PREPARATION OF A SMEAR AND HEAT FIXING
1. Using a sterilized inoculating loop, transfer loopful of liquid suspension containing bacterial to a slide or transfer an isolated colony from a culture plate to a slide with a water drop.
2. Disperse the bacteria on the lop in the drop in the drop of water on the slide and spread the drop over an area the size of a dime. It should be a thin ,even smear.
3. Allow smear to dry thoroughly.
4. Heat –fix the smear Cautiously by passing the underside of the slide through the burner flame two or three times. It fixes the cell in the slide. Do not over heat the slide as it will distort the bacterial cells.

1. Flood slide with crystal (or gentian) violet- 60 seconds.
2. Flood with Gram’s iodine – 180 seconds.
3. Carefull y decolorize with 95% ethanol until thinnest parts of the smear are colorless. (Wash with water).
a. This third step is the most critical and also the one most affected by technical variations in timing and reagents.
4. Flood with safranin (pink color) (10% Fuchsine) – 60 seconds. (Wash with water).
5. Air dry, or blot with absorbent paper.
Results
As shown below, organisms that retain the violet-iodine complexes after washing in ethanol stain purple and are termed Gram-positive, those that lose this complex stain red from the safranin counter stain are termed Gram – negative.
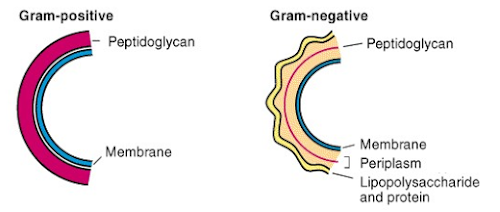
Relationship of Cell Wall Structure to the Gram Stain
In the Gram stain, an insoluble crystal violet-iodine complex is formed inside the cell, and this complex is extracted b y alcohol from gram-negative but not from gram-positive Bacteria.
The alcohol dehydrates Gram_positive Bacteria, which have very thick cell walls consisting of several layers of peptidoglycan.
This causes the pores in the walls to close, preventing the insoluble crystal violet-iodine complex from escaping.
In gram-negative Bacteria, alcohol readily penetrates the lipid-rich outer layer, and the thin peptidoglycan layer also does not prevent solvent passage, thus, the crystal violet-iodine complex is easily removed.