Drying

Learning objectives
At the end of this lecture student will be able to:
• Outline the industrial applications of drying
• Explain the differences between evaporation and drying
• Describe the stages of rate of drying
• Explain the process of drying
• Explain the stages of drying process
• Enlist types of dryers
• Discuss the construction and working process of tray dryer and vacuum dryer
• Discuss the construction and working process of fluidized bed dryer
• Explain the process operation of drum dryer
• Discuss the construction and working process of spray dryer
• Explain the process operation of freeze dryer
Drying or Dehydration
• It is the removal of all or most of the liquid by supplying latent heat to cause thermal vaporization
• Also defined as “the removal of small amounts of water or other liquid from a material by the application of heat to
about 15-20 %.”
• Removal of the liquid from a material by application of heat, & is accomplished by transfer of a liquid from the surface
into an unsaturated vapor phase
• It also includes removal of volatile liquids or water from another liquid or gas or a Suspension
• Drying is possible when the environment is unsaturated with the water vapour. Hence humidity is an important determinant for drying of solids.
• Most pharmaceutical materials are not completely free from moisture but contain some residual water, which may
vary with the temperature and humidity4
• In the majority of cases the ‘liquid’ will be water but volatile solvents such as isopropanol may also need to be
removed in a drying process.
• The product obtained from an evaporator is either concentrated solutions or suspensions or a wet slurry, whereas that
from a dryer is substantially dry solid.
• The process of evaporation involves removal of much more quantity of liquid per hour than in drying.
• Drying and Evaporation are distinguishable by relative quantities of liquid removed from the solid
Difference between Drying and Evaporation
| Sl. No. | Drying | Evaporation |
| 1. | The main operation usually carried out on solid materials e.g. powders, or products |
The main operation usually carried out on liquid materials, e.g. solution, or products |
| 2. | It means the removal of relatively small amounts of water from solids | It include the removal of large amounts of water from solutions. |
| 3. | It means the removal of water by boiling a solution. | It involves the removal of water at temperatures below its boiling point |
| 4. | Water is usually removed by circulating air over the material in order to carry away the water vapour | Water is removed from the material as pure water vapour mixed with other gases. |
Methods of drying
1. Thermal drying – with application of heat
2. Non thermal drying – without application of heat (freeze drying)
Extraction
• The extraction of liquid from solid by use of solvent
Adsorption
• By use of desiccants such as anhydrous calcium
Expression
• The expression of solid to remove liquid
• e.g. Squeezing of wetted sponge
Desiccation
Desiccation of moisture from a solid by placing it in a sealed container with a moisture-removing material
e.g. silica gel in bottle.
Applications of Drying
1. Preparation of bulk drugs
Ex. Dried aluminium hydroxide Spray dried lactose Powdered extracts
2. Improved characteristics
Drying produces material of spherical shape, uniform Size, free flowing & enhanced solubility.
e.g. Granules are dried to improve the fluidity & Compression characteristics.
3. Stability enhancement
By removal of moisture significantly reduces rate of chemical reactions, chances of microbial attack or enzymatic actions
and thus improves stability.
4. Preservation of drug product
Drying is necessary to avoid deterioration.
1. Crude drugs of animal & vegetable origin, synthetic & semi synthetic drugs, aspirin & penicillin’s tablets are undergo
chemical decomposition process.
2. Blood products, skin & tissue undergo microbial decomposition.
5. Improved handling
Removal of moisture makes the material light in weight & reduces the bulk thus the cost of transportation will be less &
storage will be efficient.
Theory of Drying
• The rate of evaporation is related to rate of heat transfer by the equation: (Temperature differential)
dW/dø = q/λ ——❶
Where,
dW/dø = rate of evaporation
q = rate of heat transfer
λ = latent heat of vaporization of water
• The rate of diffusion of moisture into air is expressed by rate equations as: (Humidity differential)
dW/dø = K ́A(Hs-Hg) ———❷
Where,
dW/dø = rate of diffusion
k ́= coefficient of mass transfer (it is not constant, but varies with velocity of passing air stream)
A = area of evaporating surface
Hs = abs. humidity at evaporating surface
Hg = abs. humidity at passing air stream
• After initial period of adjustment, the rate of evaporation is equal to rate of diffusion of vapor and rate of heat
transfer…
dW/dø = q/λ = K ́A(Hs-Hg) ——❸
• If overall heat transfer, q , is expressed as sum of rates of heat transfer by conduction, convection and radiation,
equation 3 is expanded to form :
dW/dø = (qc + qk + qr)/λ = K ́A(Hs-Hg) —–❹
rate of drying can be accelerated by increasing any of the individual terms in equation 4
In a wet solid mass, moisture is present in two forms;
a) Bound moisture
It is the minimum water (moisture) held by the material that exerts an equilibrium vapor pressure less than the pure
water at the same temperature.
Substances containing bound water are often called hygroscopic substances.
b) Unbound moisture
It is the amount of water held by the material that exerts an equilibrium vapor pressure equal to that of pure water at
the same temperature.
• Unbound water exists mostly in the voids of the solids. Thus, in a non-hygroscopic material, all the liquid is unbound
water.
• In a hygroscopic material, the unbound moisture is the liquid in excess of the equilibrium moisture content.
Measurement of the moisture in a wet solid is referred as moisture content, or MC:
Weight of dry sample
% MC =————————————-X 100
Weight of water in sample
1. Hygroscopic materials Water in: Fine capillaries Cell & fibre wall Physical interaction Contains Bound Water Condition
Vapour pressure of wet solids is less than the vapour pressure of pure water
2. Non – hygroscopic materials Water in void spaces Contains Unbound Water Condition Vapour pressure of wet solids
is equal to vapour pressure of water
Mechanism of Drying
Drying involves two processes –
Heat Transfer
It takes place from the heating medium to the solid; except in high frequency electric drying, where heat is generated
within the solid & flows to exterior surface.
Mass Transfer
It involves movement of the moisture to the surface of the solid & its subsequent evaporation from the surface. The
transfer of vapors from the surface to the surrounding is affected by external conditions like temperature, humidity, air
flow rate, pressure & evaporating surface exposed.
Equilibrium Moisture Content (EMC)
It is the amount of water present in the solid which exerts a vapor pressure equal to the vapor pressure of the
atmosphere surrounding it.
EMC at which the material is neither gaining nor losing moisture. The value of the EMC depends on the material and the
relative humidity and temperature of the air with which it is in contact.

Depending upon temperature and humidity conditions, solids may absorbs or lose moisture.
Desorption
When air is continuously passed over the solid containing moisture more than EMC, then solid loses water continuously
till EMC is reached.

Sorption
When air is continuously passed over the solid containing moisture less than EMC, then solid absorbs water continuously
till EMC is reached

Measurement of EMC
• The solid samples are placed in a series of closed chambers such as desiccators.
• Each chamber consist of solutions which maintain a fixed relative humidity in the enclosed air spaces.
• Solid samples are exposed to several humidity conditions, the exposure is continued until the material attains a
constant weight.
• The difference in the final & initial weights gives the moisture content.
Factors Affecting EMC
Nature of material
• Nonporous insoluble solids have an EMC zero EX. Talc
• For fibrous or colloidal organic substances, EMC values are high.
• For porous solids, EMC values are much higher & variable.
Nature of Air
• For air of zero humidity, EMC of all materials is zero.
• As the temperature of air increases, the EMC of solid decreases.
Free Moisture Content (FMC)
FMC is the amount of water that is free to evaporate from the solid surface.
Free moisture content (FMC) = total water content – EMC
The moisture present in the solid can be expressed on a wet weight or dry weight basis.
Mass of water in sample (kg)
% Loss on Drying (LOD) = —————————————-× 100
Total mass of wet sample (kg)
Loss on Drying (%) = initial weight of sample – weight of sample ofter drying x 100 / Initial weight of sample
Mass of water in sample (kg)
% Moisture Content (MC) =—————————————-× 100
Mass of the dry sample (kg)
Behavior of Solids during Drying or Rate of Drying
The rate of drying of a sample can be determined by suspending the weight material on a balance in a drying cabinet
and measuring the weight of sample as it dries as a function of time.
Weight of water in sample (kg)
Drying Rate = ———————————————-
Time (h) × weight of dry solid (kg)
DRYING RATE CURVE
It is obtained by plotting a graph of FMC on X- axis & drying rate on Y- axis.
Depending on external conditions & internal mechanism of fluid flow, solids show different drying patterns.
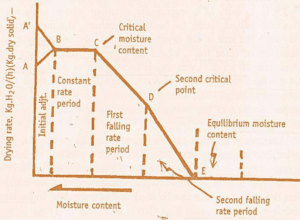
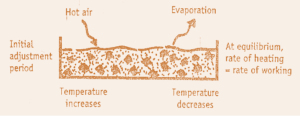
1. Initial Adjustment Period
It is time corresponding to AB curve (Heating Up Period).
During this period, solids absorbs heat & temperature is increase i.e. An wetted substance when kept for drying it
absorbs heat from surrounding & vaporization of moisture takes place which cools the surface.
Heat flows to the cooled surface at higher rates, leads to rise in temperature & evaporation again.
This continues & after some time heating & cooling rate becomes equal. This temperature is equal to wet bulb
temperature of drying air & is referred by the point B on graph.
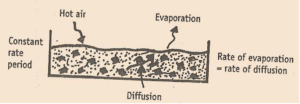
2. Constant rate period
It is time corresponding to BC curve in graph. The temperature remains constant & rate of drying is constant. During this
period, there is a continuous liquid film over the surface of solid. Moisture evaporating from the surface is replace by the
water diffusing from the interior of the solid. Also the drying rate remains constant as show in curve by region BC.
Rate of diffusion = Rate of evaporation
As drying proceeds, the coarse capillaries are completely depleted of water & solid fails to maintain uniform film. The
area over which moisture film is not present is known as ‘dry spot’.
Such dry spot start appearing & drying rate start falling & point (point C) at which decrease in drying rate start is referred
to as the critical moisture content (CMC)
3. First falling Rate period
Also known as Period of Unsaturated surface drying.
It is time corresponding to CD curve.
During this period, surface water is no longer replaced at a rate fast enough to maintain a continuous film on the
surface.
Dry spots begin to appear & rate of drying begins to fall off. The point D is referred to as second critical point.

4. Second falling rate period
It is time corresponding to DE in graph. During this Period, rate of drying falls even more rapidly than the first falling rate & no film is present on surface.
At the end, the drying rate becomes zero & moisture content of solids at this point (Point E) referred to as Equilibrium
Moisture Content (EMC).
EMC is defined as, the mass of water per unit mass of dry solid when drying limit has been attained by use of air at any
given temperature and humidity.

Factors affecting drying process
• Type, variety and size of materials
• Air temperature
• Air Humidity
• Air velocity
• Exposure time
• Initial moisture content
• Depth of drying layer
• Cost of the equipment
CLASSIFICATION OF DRYERS
Static bed dryer

Systems in which there is no relative movement among the solid particles being dried, although there may be bulk
motion of the entire drying mass. e.g: Tray dryer, Freeze dryer
Moving bed dryer
System in which the drying particles are partially separated so that they flow each other. E.g.: Drum Dryer
Pneumatic bed dryer
Systems in which the solid particles are partially suspended in an upward moving heated gas system. E.g.: Spray dryer

Fluidized bed dryer
System in which drying particles are entrained & conveyed at a high velocity gas stream.

Tray dryer
Principle
In the tray dryer, hot air is continuously circulated. Forced convection takes place to remove moisture from the solids
placed in trays. Simultaneously, the moist air is removed partially. (Shelf, cabinet or compartment dryer)
Construction
• It consist of rectangular chamber whose walls are insulated. Trays are placed inside the heating chamber. Laboratory
dryers contains 3 trays minimum & in industry 20 trays
• Each tray is rectangular or square & about 1.2 to 2.4 meters square in area. Trays are usually loaded from10 to 100
mm deep
• Distance between bottom of upper tray & surface of the substance loaded in the subsequent tray must be 40mm.

Dryer is fitted with a fan for circulating air over the trays in the corner of chamber, direction vanes are placed to direct
air in the expected path.
Working
• Wet solids is loaded into trays. Fresh air is introduced through inlet, which passes through the heaters & gets heated
up.
• Hot air is circulated by means of fans at 2 to 5 meter per second. Turbulent flow lowers the partial vapour pressure in
the atmosphere & also reduces the thickness of air boundary layer. As water evaporates from the surface, the water
diffuses from the interior of solid by capillary action. These occur in single pass of air.
• The time of contact is short & amount of water is picked up in a single pass is small. Therefore discharged air to the
tune of 80 – 90 % is circulated back through fans only 10-20% of fresh air is introduced.
Advantages
1. Handling of materials can be done without losses
2. It can be operated batch wise, for following reasons:
a) Each batch can be handled as separate entity
b) Equipment is readily adjusted for use in drying of variety of materials
c) Valuable products can be handled efficiently
Disadvantage
1. It requires more labour to load & unload
2. Time consuming
Uses
Following substances can be dried;
1. Sticky materials
2. Plastic substances
3. Precipitates
4. Chemicals
5. Tablet granules & powders
Vacuum dryer
Principle
In vacuum dryer, material is dried by the application of vacuum.
When vacuum is created, the pressure is lowered so that water boils at a lower temperature. Hence, water evaporates
faster. The heat transfer becomes efficient i.e. rate of drying enhances substantially.
Construction
• It is made up of a cast iron heavy jacketed vessel. It is so strong that it can withstand high vacuum within the oven &
steam pressure in the jacket.
• The enclosed space is divided into a number of portions by means of 20 hollow shelves, which are part of jacket. These
shelves provide large area for conduction of heat.
• Over the shelves, metal trays are placed for keeping the material
• The oven door can be locked tightly to give an air tight seal. Oven is connected to a vacuum pump by placing
condenser in between
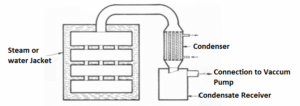

Working
• Material to be dried is spread on trays. Trays are placed on the shelves
• Pressure is decreased up to 30 to 60 kilopascals by means of vacuum pump. Door is closed firmly
• Steam or hot air is supplied into the hollow space of jacket & shelves. Heat transfer by conduction takes place
• At this vacuum, evaporation of water from the material takes place at 25-30°C, on account of lowering of boiling point.
• Water vapour passes into the condenser where condensation takes place
Advantages
1. Large surface area for heat transfer
2. Handling of material, trays & equipment is easy
3. Easy to switching over to next material
4. Hot water of desired temperature can be supplied
5. Electrically heated hollow shelves can be used
Disadvantages
1. Heat transfer coefficient are low
2. Limited capacity & used for batch process
3. More expensive than tray dryer, Labour & running cost is also high
4. There is danger of overheating as the material is in contact with steam heated surface for longer period
Uses
1. Heat sensitive materials, which undergo decomposition
2. Dusty & hygroscopic material
3. Drugs containing toxic solvents can be separated into closed containers.
4. Feed containing valuable solvents. These are recovered by condensation
5. Drugs which required as porous end products
Fluidised Bed Dryer
Principle
Hot air is passed at high pressure through a perforated bottom of container containing granules to be dried
The granules are lifted from the bottom & suspended in the stream of air. This condition is called Fluidized state
The hot gas surrounding every granule to completely dry them
Thus, materials or granules are uniformly dried.
Construction
Two types of bed dryers are available:
1. Vertical fluid bed dryer
2. Horizontal fluid bed dryer
• The dryer is made up of stainless steel or plastic. A detachable bowl is placed at the bottom of the dryer, which is used
for charging & discharging
• The bowl has perforated bottom with a wire mesh support for placing materials to be dried. A fan is mounted in the
upper part of circulating hot air
• Fresh air inlet, prefilter & heat exchanger are connected serially to heat the air to the required temperature. The temp.
of hot air & exit air are monitored
• Bag filters are placed above the drying bowl for the recovery of fines
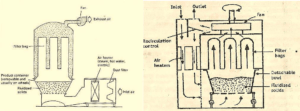
• The wet granules to be dried are placed in the detachable bowl & it is pushed into dryer
• Fresh air is allowed to pass through a prefilter, which subsequently gets heated by passing through a heat exchanger.
The hot air flows through the bottom of the bowl. The air velocity is gradually increased
• When the velocity of the air is greater than settling velocity of granules, the granules remain partially suspended in the
gas stream
• The granules rise in the container because of high velocity gas & later fall back in a random boiling motion. This
condition said to be Fluidised State.
• The gas surrounds every granule to completely dry them. The air leaves the dryer by passing through bag filters
• The entrained particles remain adhered to the inside of the bags. Periodically the bags are shaken to remove the
entrained particles
Advantages
1. It requires less time to complete drying i.e. 20 to 40 min. compared to 24hr of tray dryer
2. Different sizes with different drying capacity from 5 to 200 kg/hr
3. Handling simple & low labour cost
4. Thermal efficiency is 2 to 6 times than tray dryer
5. Mixing efficiency is also high as compared to other dryers
6. Hot spots are not observed in the dryer
7. It facilitates the drying of thermolabile substances, as contact time is short
8. It can be used for batch type or continuous type
9. It has high output from a small floor space
10. The free movement of individual particles eliminates the risk of soluble material migrating as may occur in static bed
Disadvantages
1. Many organic powders develop electrostatic charge during drying. To avoid this, efficient earthing of dryer is essential
2. The turbulence of the fluidized state of granules may cause attrition of some materials resulting in the production of
fines. but using a suitable binding agent this problem can be solved
3. Fine particles may become entrained & must be collected by bag filters
Uses
1. It is popularly used for drying of granules in the production of tablets
2. It can be used in three operations like mixing, granulation & drying
3. It is modified for coating of granules
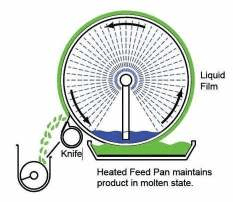
Drum drier
Principle
A heated hallow metal drum rotates on its longitudinal axis, which is partially dipped in the solution to be dried.
The solution is carried as a film on the surface of the dryer & Dried to form a layer. A suitable knife scraps the dried material, while drum is rotating
Construction
• It consist of a horizontally mounted hollow steel drum of 0.6 – 3.0 meters diameter & 0.6 – 4 meters in length, whose
external surface is polished.
• Below the drum, feed pan is placed in a such way that the drum dips partially into the feed.

• On one side of the drum a spreader is placed & on other side a doctor’s knife is placed to scrap the dried material
• A storage bin is placed connecting the knife to collect the material
Working
• Steam is passed inside the drum. Drying capacity is directly proportional to the surface area of the drum. Heat is
transferred by conduction to the material
• Drum is rotated at a rate of 1- 10 revolutions per minute. The liquid material present in the feed pan adheres as a thin
layer to the external surface of drum during its rotation
• Material is completely dried during its journey in slightly less than one rotation. the dried material is scrapped by knife,
which then falls into a storage bin. The time of contact of the material with the hot metal is 6-15 seconds only

Advantages
1. Drying time is less only few seconds. Hence, heat sensitive material can be dried
2. Less space as compared to spray dryer
3. Rates of heat & mass transfer are high
4. Product obtained is completely dried & is in the final form
Disadvantages
1. Maintenance cost is higher than spray dryer
2. Skilled operators required
Uses
It is used for drying solutions, slurries, suspensions etc. & also drying of products like;
1. Milk product
2. Starch product
3. Ferrous salt
4. Suspensions of zinc oxide & kaolin
5. Antibiotics
6. Yeast & pigments
7. Malt & glandular extracts
8. Insecticides
9. Calcium & barium carbonates
Spray Dryer
Principle
The fluid to be dried is atomized into fine droplets, which are thrown radially into a moving stream of hot gas.
Temperature of droplets is immediately increased & fine droplets get dried in the form of spherical particles. This
process completes in few seconds before the droplets reach the wall of dryer
Construction
It consist of a large cylindrical drying chamber with a short conical bottom, made up of stainless steel. Diameter of 2.5-
9.0 meters & height 25.0 meters or more
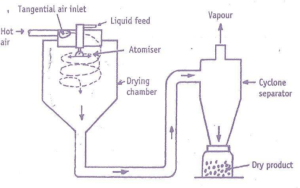
• An inlet for hot air is placed in the roof of the chamber & another inlet carrying spray disk atomizer is set in the roof.
• Spray disk atomizer is about 300 mm in dia. & rotates at a speed of 3000 to 50,000 rpm. Bottom of the dryer is
connected to a cyclone separator.
Working
Drying of material in spray dryer involves three stages;
a) Atomization of liquid
The feed is introduced through the atomizer either by gravity or by using suitable pump to form fine droplets. The
properties of final product depends upon the droplet form. Atomizer can be of any type:
1. Pneumatic atomizer
2. Pressure nozzle type
3. Spinning disc atomizer
Rate of feed is adjusted in a such a way that droplets should be completely dried before reaching walls of drying
chamber.
b) Drying of liquid droplets
• Surface of a liquid drop is dried immediately to form a tough shell. Liquid inside must escape by diffusing through the
shell at a particular rate
• Heat transfer from outside to inside takes place at a rate greater than liquid diffusion rate. As a result, heat inside
mounts up which allow the liquid to evaporate. This leads to increase in internal pressure, which causes droplets to swell
• The shells thickness decreases where as permeability for vapour increases. If the shell is neither elastic nor permeable
it ruptures.
c) Recovery of dried product
• Centrifugal force of atomizer drives the droplet to follow helical path. Particles are dried during their journey & finally
fall at the conical bottom
• All these processes are completed in a few seconds. Particle size of the final product ranges from 2 to 500 μm
• Particle size depends upon solid content of the feed, liquid viscosity, feed rate & disc speed
• Capacity of spray dryer – 2000 kg / h.
Advantages
1. It is continuous process & drying completes within 3 to 300 sec
2. Labour cost is low
3. Product of uniform & controllable size can be obtained
4. Fine droplets form provide large surface area for heat & mass transfer. Product shows excellent solubility
5. Either solution or suspensions or thin paste can be dried in one step to get final product ready for package
6. Drying of sterile product & Reconstituted product
7. Globules of an emulsion can be dried with the dispersed phase inside & layer of continuous phase outside
Disadvantages
1. It is very bulky & expensive
2. Such huge equipment is not always easy to operate
3. The thermal efficiency is low, as much heat is lost in the discharged gases
Uses
Spray dryers are used compulsorily, if:
1. The product is a better form than that obtained by any other dryer
2. The quantity to be dried is large
3. The product is thermolabile, hygroscopic or undergo chemical decomposition
A few product that are dried using spray dryer are: Citric acid, Calcium, Sodium, Gelatin. sulphate
Freeze Dryer
It is also known as lyophilization i.e. system is made solvent loving for removing the same.
Principle
• In freeze drying, water is removed from the frozen state by Sublimation i.e. direct change of water from solid into
vapour without conversion to liquid phase.
• Solid-liquid- vapour equilibrium phase diagram of water is useful to decide the experimental conditions. The drying is
achieved by subjecting material to temperature & pressure below the triple point.
• Under this conditions, any heat transferred is used as latent heat & ice sublimes directly into vapour state.
• The water vapour is removed from the system by condensation in a cold trap maintained at a temperature lower than
frozen material.

Construction
Freeze dryer consist of
• Drying chamber in which trays are locked
• Heat supply in the form of radiation source, heating coils
• Vapour condensing or adsorption system
• Vacuum pump or steam ejector or both

Working
The working of freeze dryer consist of following steps;
1. Preparation & pretreatment
The volume of solution introduced into the container is limited by its capacity. Therefore pretreatment is essential. The
solutions are preconcentrated under the normal vacuum tray drying. This reduces the actual drying by 8 to 10 times.
2. Prefreezing to solidify water
Vials, ampoules or bottles in which the aqueous solution is packed are frozen in cold shelves (- 50oC).
The normal cooling rate is about 1 to 3 Kelvin/ minute so that large ice crystals with relatively large holes are formed on
sublimation of ice. This is also responsible for giving a porous product.

3. Primary Drying
• It means sublimation of ice under vacuum. The temperature & pressure should be below the triple point of water i.e.
0.0098oC & 4.58 mmHg for sublimation, when water is alone present
• When a Solution of a solid is dried, the depression of freezing point of water occurs. Hence, it is essential that the
temperature be brought below the eutectic point. The pressure & temp. at which the frozen solid vaporizes without
conversion to liquid is referred to as the eutectic point. Depending on the drug substances dissolved in water, the
eutectic point is determined. The usual range is from -10oC to -30oC.
The conditions of 1 to 8 K below eutectic point is sufficient.
• Vacuum is applied to the tune of about 3 mmHg on the frozen sample and the temperature is linearly increased about
30 oC in a span of 2 hrs. Heat (About 2900 kilojoules/ Kg) is supplied which transfer as latent heat & ice sublimes directly
into vapour state. As the drying proceeds, thickness of dried solids increases. Primary drying stage removes easily
removable water, about 98% to 99%.
4. Secondary Drying
It is removable of residual moisture under high Vacuum. The temp. of solid is raised to as high as 50 to 60oC but vacuum
is lowered below that is used in primary drying.
The rate of drying is very low & it takes about 10 to 20 hrs
5. Packing
After vacuum is replaced by inert gas, the bottles & vials are closed
Advantages
1. Thermo labile substances can be dried
2. Denaturation does not occur
3. Migration of salts & other solutes does not take place
4. Moisture level can be kept as low as possible
5. Product is porous & uniform
6. Sterility can be maintained
7. Material can be dried in its final container such as single dose & multiple dose
Disadvantages
1. The product is prone to oxidation, due to high porosity & large surface area. Therefore, the product should be packed
in vacuum or using inert gas.
2. Equipment & running cost is very high.
3. The period of drying is very high. Time cannot be shortened.
4. It is difficult to adopt the method for solutions containing non-aqueous solvents.
Uses
It is used for drying of number of product;
1. Blood plasma & its fractionated product
2. Bacterial & viral culture
3. Antibiotics & plant extracts
4. Steroids, vitamins & enzymes
Summary
• Drying is the process of complete removal of solvent from the substance and the end product is a solid
• Thermal and non-thermal are the two methods of drying process Mass and heat trasfer are the two process of drying
• Wet solid mass contains both bound and unbound water
• Equilibrium moisture content is the amount of water present in the solid which exerts vapor pressure equal to
atmosphere surrounding it
• Rate of drying curve is obtained by plotting free moisture content versus drying rate
• Different stages of drying include initial adjustment period, constant rate period, first falling rate period and second
falling rate period
• Tray dryer is used for localized drying of substances
• Vacuum dryer is used for drying heat sensitive substances
• Fluidized bed dryer is used for generalized drying process
• Bag filters used in the F.B.D. prevents the escape of finer particles from the equipment during drying process
• Drum dryers are used for drying solutions, slurries and suspensions
• Spray dryer utilizes generalized drying process
• It can be used for coating of polymer over the granules – Spansule technology
• Freeze dryer utilizes triple point as the drying phenomenon
• It can be used in the preparation of vaccines and blood products
For Detailed PDF Notes Click on Download Button
