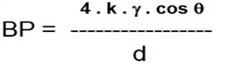Validation of sterilization
Contents
• Validation – meaning and
significance
• Sterilization indicators
– Physical indicators
– Chemical indicators
– Biological indicators
Intended learning objectives
At the
end of this lecture, the student will be able to:
• Explain the significance of
validation in sterilization
• List the sterilization indicators
• Describe the working of
sterilization indicators
Validation- History
•
The concept of validation was first proposed by
two Food and Drug Administration (FDA) officials, Ted Byers and Bud
Loftus, in the mid-1970s in order to improve the quality of pharmaceuticals
•
It was proposed in direct response to several
problems in the sterility of large volume parenteral market
•
The first
validation activities were focused on the processes involved in making sterile
products, but later on were applied for
associated processes such as
environmental control, media fill, equipment sanitization and
purified water production
Validation of sterilization
• Validation means demonstrating that a process
will consistently produce the results that it is intended to
• Validation is
a process of establishing documentary evidence demonstrating that a procedure,
process, or activity carried out in production or testing maintains the desired
level of compliance at all stages.
• Validation would be necessary for
each of the individual aspects of the manufacturing process,
• Environmental monitoring
• Raw materials quality assessment
• Sterilization process itself
• The sterility testing procedure
Sterilization indicators
Sterilization procedures should
be monitored through a combination of physical, chemical, and biological
techniques designed to evaluate the sterilizing conditions and the
procedure’s effectiveness
• Physical indicators
• Chemical indicators
• Biological indicators
Physical indicators
Observing
the gauges or displays on the sterilizer for
• Assessing the cycle time
• Temperature
• Pressure of sterilization equipment
• Relative humidity
Heat
sterilization
• A temperature record chart is made –
part of the batch documentation
• Compared against a master
temperature record (MTR)
• Information on heat distribution and
penetration within a sterilizer can be gained by the use of thermocouples
Gaseous
sterilization
• Measurement of elevated temperatures
using temperature probes
• Routine leak tests to ensure
gas-tight seals
• Pressure and humidity measurements
are recorded.
• Gas concentration is measured, often
by reference to weight of gas used
Radiation
sterilization
• A plastic (often perspex) dosimeter
is used
• It
gradually darkens in proportion to the radiation absorbed
Integrity testing sterilizing filters
• Membrane
filters have been used successfully for many years to remove yeast, bacteria
and particulate from fluid streams
• To
test the integrity of filters, the following tests are done:
a) Destructive
test
b) Non
– destructive integrity test
a) Destructive test- Destructive challenge
testing is the best way to determine a sterilizing filter’s ability to retain
bacteria. During the bacterial retention test, 0.22 µm filter discs and devices
are challenged with a solution of culture medium containing bacteria (Brevundimonas
diminuta ATCC 19146) at a minimum challenge of 107 per cm2.
The effluent is then passed through a second 0.45 µm filter disc that is placed
on an agar plate and incubated.
– The filter cannot
be used for filtration purposes again
b) Non – destructive integrity test, may be
done on filters before and after filtration process
-This is done to ensure that the filter meets
specification, is properly installed and intact during filtration, and to
confirm the rating of the filter
– Integrity testing before filtration process monitors
filter integrity, preventing use of a non-integral filter.
– Integrity testing after a batch has been filtered can detect if the integrity
of the filter has been compromised during the process
There are 3 major tests used to determine the integrity of a
membrane filter: the Bubble Point Test, the Forward Flow Test, and the Pressure
Hold Test
• All three tests are based on the same physics,
the flow of a gas through a liquid-wetted membrane under applied gas pressures
• They differ in which part of the flow/pressure
spectrum they examine
• This
is an indirect method for the measurement of pore size of filter
Bubble
point pressure test
• Bubble
point test is based on the fact that
liquid is held in the pores of the filter by surface tension and capillary
forces
• The minimum pressure ( as seen on the pressure
dial) required to force liquid out of the pores is a measure of the pore
diameter
The bubble point is expressed as :
Where
k = shape correction factor
Ύ = surface tension
cos ɵ = liquid solid contact angle
d = pore diameter
• Consists
of immersing the filter candles in water or filling the funnel with the liquid
in the case of a membrane filter or sintered glass filter
• Air
or gas is passed from the bottom of the filter
• The
pressure of Air/ gas is gradually increased until the first bubble is seen at
the filter/liquid interface
Filtration
sterilization
• Bubble point pressure test
Chemical indicators
• Chemical
indicators are designed to respond to a characteristic change to one or more of
the physical conditions within the sterilizing chamber
• The
principle is based on the ability of heat, steam, gases and ionization
radiation to alter the chemical and/or physical characteristics of chemical substances
• Change should take place only when
satisfactory conditions for sterilization prevail
• Used
for monitoring a sterilization process
• Chemical indicators generally undergo
melting or colour changes
• Included in strategically placed
containers or packages
• Monitors the conditions prevailing at the coolest or
most inaccessible parts of a sterilizer
Bowie
Dick test: Organic
chemical in a printing ink base impregnated into a carrier material. A
combination of moisture and heat produces a darkening of the ink
Brown’s
tube: Sealed tubes
partly filled with a solution which changes colour at elevated temperatures;
rate of colour change is proportional to temperature
Thermalog
S: Consists of a
blue dye in a waxy pellet. At autoclaving temperatures, and in the continued
presence of steam, the pellet melts and travels along a paper wick forming a
blue band the length of which is dependent upon both exposure time and
temperature
Thermalog G: Same as Thermalog S. Used for gaseous
sterilization
Biological indicators
• Consist of standardized bacterial
spore preparations
• Usually in the form either of
suspensions in water or culture medium or of spores dried on paper, aluminium
or plastic carriers.
• As with chemical indicators, they
are usually placed in dummy packs located at strategic sites in the sterilizer
• After the sterilization process, the
aqueous suspensions or spores on carriers are aseptically transferred to an
appropriate nutrient medium, which is then incubated and periodically examined
for signs of growth.
• The bacterial species to be used in
a BI must be selected carefully, as it must be non-pathogenic and should
possess above-average resistance to the
particular sterilization process.
Filtration
sterilization
• Measuring the ability of a filter to
produce a sterile filtrate from a culture of a suitable organism
• Serratia marcescens
has been
used for filters of 0.45mm pore size,
• Brevundimonas diminuta (formerly Pseudomonas diminuta) having a minimum dimension of 0.3mm is applied to filters of
0.22mm pore size.
Summary
• Validation means demonstrating that
a process will consistently produce the results that it is intended to
• Sterilization indicators – Physical
indicators, Chemical indicators, Biological indicators
• Physical indicators validate the
temperature, pressure, radiation dose or filter pore size
• Chemical indicator – Based on the
ability of heat, steam, sterilant gases and ionizing radiation to alter the
chemical and/or physical characteristics of a variety of chemical substances
• Biological indicators – Consist of
standardized bacterial spore preparations